GVS Group is one of the world's leading manufacturers of filter solutions, was established in 1979, throughout 45+ year history, GVS has evolved from a supplier of components for the healthcare sector to a global Group that provides a range of diversified, high-tech filtration solutions.
GVS has always dedicated considerable attention to research, development and innovation in its product lines and production processes, constantly improving its ability to offer solutions that effectively support the needs of its customers.
GVS strong focus on internationalization has led to the opening of 18 production plants located in Italy, UK, Brazil, USA, China, Mexico and Romania, as well as 30 sales offices in China, USA, Turkey, Argentina, Malaysia, Japan Korea.
Wide range of products and custom design expertise
GVS produces a wide range of filter materials, filters and off-the-shelf components in all its divisions, enabling its customers to reduce the design time for new product launches.
All the GVS divisions work in highly regulated environments and the Group therefore operates with extremely high quality standards.
Thanks to its research and development centres located all over the world, GVS is also able to offer an extremely efficient and personalised service to meet its customers’ needs: from product conception and design to testing and mass production.
Dynamic and flexible structure
GVS has developed a streamlined, dynamic and technologically advanced structure that has made it possible to achieve constant and balanced growth. The Group currently employs a total of 2,500 people who work in automated assembly departments, in lines for the production and processing of filter membranes and in class 10,000 and 100,000 cleanrooms.
Global growth
Since its foundation, the GVS Group has always demonstrated a strong trend towards development in global markets.
Through its corporate headquarters in Bologna and its offices abroad, GVS has established a worldwide sales and production network to closely support its customers, offering them a more effective and efficient assistance service.
In sectors that are highly technological and critical in terms of safety, GVS has considered a “glocal” approach to be vitally important. This approach offers the ability to work locally with customers, while also remaining in communication with their offices at a global level.
GVS currently has 18 plants in Italy , United Kingdom,Brazil, United States, China, Mexico and Romania, as well as 30 sales offices located in Argentina, Russia, Turkey, Malaysia, Japan, Korea and China.








GVS was founded in 1979 and initially focused on medical blood filters production. The company later branched out into the production of a wide range of proprietary products, rapidly gaining considerable success and receiving recognition worldwide. The company was founded by Grazia Valentini with the support of her father Renato, one of the first injection mold designers in Italy. Between them they developed the first innovative techniques of automatic co-molding on horizontal presses and these were the foundations on which the present company technologies were based.
The work done in critical and maximum-safety sectors such as the Automotive sector (ABS braking systems) the Medical sector (blood filtering), Life Sciences (diagnostic and biotechnology), Safety and Biohazard (respiratory and antiviral protection), Molecular Filtration (chemical protection) has encouraged GVS's inclination towards constant innovation and improvement. GVS is today considered a group with a fine tradition of quality and high technological innovation in design and materials used, and it is these features which are the key to its international success.
Throughout its history, GVS has always invested in people and benefitted from the skills, energy and enthusiasm they bring to the company. It's the people within the business that drive the success of the group. GVS recognise this and provide incentives for their creativity whilst and cultivating their professional skills to ensure continuous development and innovation.
GVS established by Ms. Grazia Valentini as GVS Srl producing filters for medical applications
Automotive division is set up and a second production plant opened.
Opening of the third production plant.
Opening of a subsidiary in Belgrade, to cover all the ex-Yugoslavia market.
- GVS obtains ISO 9001: 1994 Quality System Management Standard Certification.
- A GVS subsidiary is opened in Buenos Aires.
GVS obtains QS 9000 and AVSQ'94 Certifications.
Setting up of GVS do Brasil at Monte Mor, Sao Paolo, Brazil.
Opening of the Group's new head-offices in Bologna.
Change-over to GVS SpA, to best sustain future growth. The B Group private equity fund contributes capital through the purchase of a minority share of GVS.
Opening of a sales office in Shanghai, China.
Startup of a trading partnership in Tokyo, Japan.
Construction and opening of the fourth production plant in Italy, GVS Sud, in Manocalzati (AV).
Opening of the new branch, GVS Filter Technology Inc., in Indianapolis, USA.
Opening of a new branch in Guadalajara, Mexico.
GVS Filter Technology Inc. moves in to a new production facility in the USA.
GVS obtains ISO/TS 16949:2002 certification, and ISO13485 certification for the medical sector. GVS Shareholders buy back shares from the B Group fund
Started joint venture with a local Chinese medical company.
Brazil: Doubling of production capacity by extending the production plant.
China: 100% Acquisition of Local Chinese JV (medical).
GVS Italian headquarters successfully achieved UNI EN ISO 14001:2004 certification.
Spain: Acquisition of GEMA Medical, Laboratory Filtration.
Acquisition of Fenchurch Environmental Group in the UK.
GVS obtains ISO 9001: 2008 quality system management standard certification revision
Mandarin Capital Partners enters contributing contributing capital through the purchase of a minority share in GVS Group.
Complete reorganisation of the Chinese Suzhou plant into a world class manufacturing site.
Puerto Rico: Acquisition of IVEX and Nutrivex Filter product line of NP Medical.
Japan: Startup of GVS Japan and Nihon Nypro Acquisition
UK: Complete the acquisition of the UK Fenchurch Group
GVS acquired a majority stake in Maine Manufacturing to facilitate the acquisition of the Filter & Membrane business of GE Osmonics and GE Healthcare Lifesciences.
UK: Acquisition of 100% Remploy Ltd, HEPA filter producer (C&I Filtration Division)
USA: Bring in the production line for Osmonics, GE Healthcare。
GVS SpA: Firstbond issuing with a private placement subscriber.
Complete Start up GVS Korea & GVS Russia.
USA: Acquisition of Maine Manufacturing。
Complete Mandarin Capital Partners buy back.
3000-sqm expansion of the Italian manufactoring site.
New 8.000 sqm manufacturing plant in Romania.
Reorganisation of the UK manufacturing site.
Group launch of a new supply chain system.
100% Acquisition of a Brazilian disposable mask company for the Safety Division.
GVS complete the acquisition of SEGRE AB, swedish manufacturer of disposable masks.

Technology Research & Development
Technology is central to GVS’s business. Thanks to pioneering strategic choices, large investment in research and development, which accounted for 8% of revenues in 2019, and cutting-edge management systems, the Group has achieved considerable success and has received numerous international awards over the years.
Constantly Committed to Development
In order to offer its customers a wide range of services and products distinguished by their high quality and reliability, GVS boasts state-of-the-art equipment and technical systems to support its research and technological development programs and the design of new products. All GVS plants share these innovations at a global level, which contributes to continuous development and enables the Group to maintain its characteristic level of technological excellence.
OEM customers are supported step-by-step by the technical project team, from concept through to industrial production. Proprietary products are developed in-house and feature innovations that are technically advanced in comparison to products on the international market. This design process has resulted in a large number of patents being filed and accepted for the more unique design features within the GVS product range.
Where possible, the design teams use membranes and filtration materials produced in-house by GVS to enable the products to offer the best possible performance.
One of the fundamental drivers behind the Group’s continuous development is its constant scientific collaboration with outstanding research centres, universities and laboratories around the world.
This process has led the Group to file and register numerous patents characterised by a unique design within the GVS product range.

GVS is a quality-oriented Group and its quality procedures now represent a shared starting point for the all the collaborators of the Group, which continuously innovates and improves its structure and organisational processes, developing and monitoring a system that guarantees a high level of quality and excellence for both its internal and external customers.
Satisfaction of customer expectations and staff training are the main goals of GVS which, applying the principles of quality and continuous improvement, uses advanced design technologies, error prevention methods and control systems throughout its production process to reduce the percentage of products that do not comply with specifications and therefore to pursue the goal of 100% defect-free production.
GVS obtained its first certifications in 1995 and today the Group and its plants hold numerous certifications at a global level. 9 GVS plants have all the necessary production certifications for the Health & Safety division, whose products are certified for worldwide distribution. The main certifications in this area are EN and NIOSH, respectively used in Europe and the United States. With regard to the Energy & Mobility division, all production facilities have moved from the old QS9000 certifications to the more recent ISO-TS 16949 certifications and are also in the process of obtaining the ISO 14001 environmental certifications. Furthermore, in the Healthcare & Life Sciences area, the plants operate with ISO 13485 certifications and are FDA-approved. Many of the products in this division have the CE or 510k mark (the equivalent of the CE mark valid for the FDA in the United States), as well as national registrations for the specific countries where the Group operates.
STABLE DEVELOPMENT IN DIVERSE MARKET SECTORS
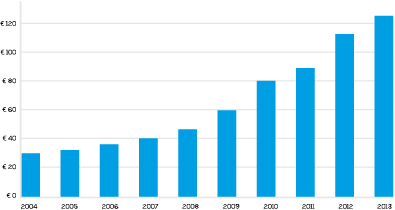
The GVS Group has witnessed a constant and significant improvement in its economic and production figures over the last 40 years. Today the business covers 6 different product groups and divisions covering a wide range of different market sectors. In 2018 the Group reached a turnover of 203 million Euro and a produced over 1.6 billion items selling into 60 countries through the Groups own commercial network.
GLOBAL PRODUCTION AND DISTRIBUTION
GVS Group's clear strategy to develop internationally by scaling existing business models into new markets has led to the opening of 12 production facilities. These are located in Italy, UK, USA, Brazil, China and Romania, as well as offices in Russia, Turkey, Argentina, Japan, Korea. The Head Office and 3 manufacturing facilities are based in Italy and produce 6% of the sales. The remaining 94% of the turnover is from main international markets: Europe, North and South America, and Asia.